Use our filter system to find the news that is more relevant to your interests:
Filter by:

September 18th 2023
Metals can have many different qualities, and strength is an important one for several industries. Metal alloys are often stronger than pure metals and there are different types of strength that vary in importance depending on what they are being used for.
Because there are a number of variables in which to categorise the strength of metal, we have listed 6 metals that are favoured for either their compressive strength, tensile strength, yield strength, and impact strength.

September 18th 2023
Titanium is a lustrous, silvery coloured metal that is known to have particularly high strength and low density. It was first discovered in Cornwall in 1791 and takes its name from Greek mythology, after the Titans.

July 27th 2022
The last four years have been quite a period for William Rowland Ltd (WR), even in the context of an almost 200 year old business that has channelled metal additives into key supply chains, through several significant global events, including the 2nd world war.

March 4th 2021
William Rowland Metal Finishing (MF) have been surface treating stainless steel and non-ferrous metals since 1940.

January 7th 2021
Welcome to WR Refined Metals, the largest and longest established segment of William Rowland. Providing a range of base metals, Ferro Alloys, Minor Metals and Noble Alloys for over 190 years spanning both world wars. WR, when trading as British Metal Corporation was a key partner supplying metals for the war effort.

October 5th 2020
Melting has enabled William Rowland to work with their partners using both Airmelt and Vacuum Induction furnaces. Specially prepared blends of revert or scrap, revert & virgin and 100% virgin materials can be cast into ingots or VIM cast stick. William Rowland also offer the service of toll melting our customer supplied revert enabling maximum value retention. In addition, William Rowland can refine the melts to achieve our customer’s bespoke specifications. William Rowlands trusted partners are also valued customers of the business, strong reciprocal “partnerships” in every sense of the word.

July 8th 2020
Following a challenge from a customer, William Rowland have successfully atomised a powder batch from a specially selected 100% revert blend of raw material. This powder is designed for use in additive manufacturing (AM).

April 5th 2020
We offer a range of Refined Metals, Revert Alloys, Metals Powders and Alloys & Solders, operating from two facilities in the UK, one in Tankersley on the border between Sheffield and Barnsley and the other in the West Midlands. Following the relocation and expansion in Q1 2020, we are now offering a range of ‘toll’ services, some at both locations. Below are some of the processes on offer at this time, please get in touch to discuss your requirements.

March 9th 2020
You can find us in our new lovely premises in Barnsley! We are pleased to announce that the relocation has now taken place following a phased relocation of equipment and most recently employees. WR is therefore now operating from the new facilities and your usual contacts and contact information remain unchanged apart from the address.

February 12th 2020
William Rowland (WR) today acquired EF Westaway (Westaways), Sheffield, UK bringing two companies with strong heritage together. The combined businesses become stronger in serving castings and forgings companies with a broader array of products and services.

August 12th 2019
Do you know what metals you are wearing?

August 5th 2019
Tin is a soft, pliable metal, with many useful applications. It is usually extracted from cassiterite, a mineral most commonly mined in the 'tin belt' region of China, Indonesia, and Thailand. While it's most widely known use is as a coating for steel cans (something its non-corrosive properties make it particularly suited to), tin can be used in a broad range of commercial and industrial contexts.

March 13th 2019
William Rowland has signed an agreement with Barmston Developments, a joint venture between Wilton Developments and Clugston Estates to acquire land and build a state of the art new manufacturing facility at Enterprise 36 in Barnsley. This will involve relocation from the company’s current Meadow Street, Sheffield premises to a new 6.5-acre site close to Junction 36 of the M1 within the Sheffield City Region.

February 5th 2019
At William Rowland, we have been providing our customers around the world with the very best in high purity metal, alloys, powders, foundry additives and more for over 180 years, making us one of the oldest metal trading companies in the world today.We have an extensive selection of metals and related products, suitable for various industries. Here are just a few of them:

November 27th 2018
Low melting point alloys (sometimes referred to as fusible alloys) fulfil a number of functions throughout the manufacturing industry. Boasting a number of interesting properties unique to them, they have numerous applications for tools and components.
May 3rd 2018
A ferrous metal is made up of iron and other metals to give the ferrous properties required. Although iron on its own is often referred to as a ferrous metal, the term really relates to any metal compounds that have iron in them. To test if a metal is ferrous or not, the valance is measured. A valence of +2 means that the metal is ferrous.

February 15th 2018
Antimony is a semimetallic chemical element which can exist in two forms: the metallic form is bright, silvery, hard and brittle; the non metallic form is a grey powder. Antimony is a poor conductor of heat and electricity, it is stable in dry air and is not attacked by dilute acids or alkalis. Antimony and some of its alloys expand on cooling. Antimony has been known since ancient times. It is sometimes found free in nature, but is usually obtained from the ores stibnite (Sb2S3) and valentinite (Sb2O3). Nicolas Lémery, a French chemist, was the first person to scientifically study antimony and its compounds. He published his findings in 1707. Antimony makes up about 0.00002% of the earth's crust.
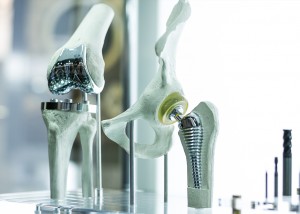
December 12th 2017
The most commonly used metals in surgery are surgical stainless steel and titanium. Surgical stainless steel is a term used to label particular grades of stainless steel that are commonly used in biomedical appliances. The most common forms are austenitic 316 stainless steel and martensitic 440 and 420 stainless steels and whilst there is no certain definition of what constitutes surgical stainless steel, manufacturers generally recognise this to be any grade of corrosion resistant steel.

December 5th 2017
Scrap metal is made up of recycled materials that have been left over from manufacturing and consumptions of products that use metals. This could be anything from parts of old vehicles, to surplus materials that have been discarded during the manufacturing process. It does not, however, include gold and silver, or any other alloy that is comprised of more than 2% of either gold or silver.

November 6th 2017
Tin is a chemical element, belonging to group 14 of the periodic table. Its symbol is Sn, which comes from the Latin stannum, and it is what we call a post-transition metal. It is commonly used in alloys, including bronze and pewter, as well as in the plating of steel to add a corrosion resistant layer.

September 26th 2017
In the society of today, recycling is more prevalent than ever. We are encouraged to recycle whenever we can, but what happens to the things we do recycle? Where do they end up?

September 15th 2017
The city of Sheffield is well known for steel manufacturing and metallurgy. From the 18th century onwards it was quickly established as one of the main industrial cities in the UK, although it was recognised for the manufacture of knives as early as the 14th century with a notable mention from Chaucuer in The Reeve’s Tale.

September 7th 2017
‘Casting’ is a process used in metalworking where a metal in liquid form is poured into a mould and allowed to cool in the cavity to form a specific shape. It is a commonly used process for making complex shapes as the use of the mould allows for great detail and it is more economical than other processes might be. Once the metal has solidified it is removed from the mould and is known as a casting. Items commonly produced by casting include pieces of jewellery, sculptures, tools and some weapons.

August 23rd 2017
Although they are undoubtedly still impressive, skyscrapers have become a common feature of modern cities that we have become accustomed to seeing on busy skylines such as New York and London. Constantly evolving, they push the limits of architectural design, with the Burj Khalifa pushing an imposing 828 metres in height. However, this is a far cry from the humble origins of the modern skyscraper which began in 1884 with a small structure of only 10 storeys in Chicago.

August 14th 2017
Applying heat to different metals can have a dramatic effect on them and can completely alter their structural, magnetic and electrical properties. There are several methods that can be used to change metals through heat, in order to enhance more favourable qualities, and the varying methods that are used will depend upon the metal and the desired result.

August 5th 2017
Galvanizing, or galvanization, is the term used for the process of applying a layer of zinc to steel or iron, in order to create a protective coating. It is commonly used as a method of protecting metals from corrosion as the layer of zinc prevents the metal from oxidizing.

July 21st 2017
Discover the best mining musuems in the UK offering a much needed insight into the history of mining.

June 21st 2017
Stainless steel is an immensely popular steel alloy that is highly prized for its resistance to corrosion and used in a variety of ways, including cutlery, medicine and jewelry. There are different types of stainless steel and the type will depend on what has been added to it. The fact that different varieties can be formed gives this an alloy an immense versatility which explains its popularity and suitability for a wide number of applications.

June 10th 2017
As Donald Trump increases protectionism in the United States, UK steel workers have voiced their concerns and begun lobbying the government in an effort not to be cut off from the American market.

May 18th 2017
Welding is the process of joining together materials, usually metals, but fusing them together through melting the surface points. Two pieces of metal can then be joined to form a single piece. Welding is used for all sorts of reasons from creating metal structures such as bridges and sculptures, to repairing damaged metal structures such as ships

January 17th 2017
Additive Manufacturing (AM), also known as 3D printing, rapid prototyping or freeform fabrication is a process in which layers of material are used to form three dimensional objects under computer control; opposed to Subtractive Manufacturing such as machining.

January 10th 2017
Tin mining in Britain has been occurring since before the Bronze Age – as tin is an essential component to smelt bronze there could not actually have been a Bronze Age without it. Two of the main areas for this were Cornwall and Devon, both of which provided most of the tin, copper and arsenic used in the UK up to the 20th Century.

December 27th 2016
Cold Casting is the term used to describe the process of mixing metal powders in a resin to create decorative features, which look as though they have been cast in metal. The castings give the appearance of solid metal, but are cheaper to produce, and are popular for use in sculptures, decorative paneling and furniture fittings, to name a few. Cold casting is also a popular technique for restoring antiques, or older furniture/household items. For example, if have something with brass fittings and you are missing one, cold casting could reproduce the part without the cost and time of doing it properly.

December 7th 2016
This week the Home Office have launched a review of the Scrap Metal Dealers Act, the latest version of which came into place several years ago in October 2013. Any stakeholders have been given until the end of January to submit ideas on possible amendments to the legislation, but what are the current rules surrounding scrap metal?

November 11th 2016
LMEprecious is a new initiative created by the London Metal Exchange (LME), the World Gold Council and a group of leading industry players, which will launch a new set of precious metal products and broaden the offering for the precious metals community.

November 3rd 2016
In the aftermath of Brexit there has been much uncertainty regarding the future of the British economy and what the impact might be on the automotive industry, which for the past decade has been one of our biggest success stories. The industry currently provides approximately 800,000 jobs across the UK, accounting for over £69.5 billion turnover last year, and adding £15.5 billion in value to economy.

October 7th 2016
Nickel chrome alloys, commonly referred to as Nichrome, stand out as versatile materials with a unique composition of nickel, chromium, and sometimes iron. The chemical formula for these alloys is NiCr, and in the presence of iron, it becomes NiFeCr. This blog post from William Rowland explores the remarkable properties and diverse applications of nickel chrome alloys, including their significance in various industries.

September 26th 2016
A non-ferrous metal can be described as any metal, including alloys, that does not contain iron (ferrite). They are more expensive than ferrous metals and have properties that include low weight, resistance to corrosion and a high level of conductivity, making them more desirable. Well known non-ferrous metals include copper, lead, zinc, aluminium, tin, nickel, gold, silver and platinum. Because non-ferrous metals are very malleable they are more suited to for use where weight is an important factor. For example, in aircraft manufacture the materials must be strong but also as light as possible. Non-ferrous metals also have a higher resistance to corrosion than ferrous metals so they are popular for things such as water pipes, roofing and road signs which are exposed to the elements.

September 7th 2016
As an indication of our commitment to the supply of metal powders, William Rowland Ltd are going to exhibit at the forthcoming World Powder Metallurgy 2016 Congress and Exhibition taking place in Hamburg, Germany from the 9th to the 13th October 2016.

August 27th 2016
The metals used in the aircraft manufacturing industry include steel, aluminium and titanium with each possessing certain qualities that make them ideal for this use. Aircraft construction demands materials that are both durable and lightweight, as well as being able to withstand severe pressure at high altitudes, and exposure to the elements.

August 17th 2016
Almost all metals can be recycled and over 400 million tonnes are recycled each year worldwide. Many metals can be melted down without compromise to their strength or durability, meaning they can be used again and again in manufacturing new products. Metal recycling is a £5.6 billion industry in the UK with both ferrous and non-ferrous metals being used to make secondary raw materials for the smelting of new metals

July 18th 2016
Historically, different precious metals have been used to make coins used for currency. Indeed, before there were standardised currency systems with coins, metals themselves were used on their own as a precious commodity representing value typically those considered precious metals such as gold and silver.

June 27th 2016
What is the most abundant metal in the world? Aluminium is the most common metal found in the Earth's crust, and the third most abundant element.
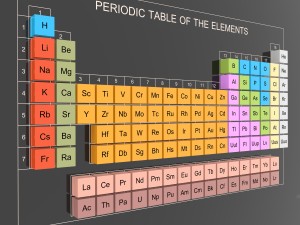
May 18th 2016
The term 'transition metals' is generally used to describe any element in the d-block of the periodic table, which includes groups 3 to 12. A transition metal is an element whose atom has a partially filled d sub shell, or which can give rise to cations with an incomplete d sub shell.

October 28th 2015
The London Metal Exchange (LME) is the world’s largest market in options and futures contracts on base and other metals.

October 21st 2015
Formerly established in 1877, the London Metal Exchange (or LME) is 138 years old. Its origins, however, can be traced as far back as the reign of Elizabeth I. In its earliest days, traders meet in London’s coffee houses to do business – the Jerusalem Coffee House being a particular favourite.

September 22nd 2015
Additive manufacturing (3D printing) is used in a variety of different industries in a variety of different ways. While not the most popular form of manufacturing, it is quickly gaining recognition for fast, simple, and low-cost manufacturing of both small run and large run components.

August 24th 2015
Everyone knows that the automotive industry uses metal. The frames of our vehicles are metal, metal is used to make the majority of the mechanical components, and is even used in the electronics. But what kinds of metals are used in the automotive industry?

August 3rd 2015
During it's now 185 year history William Rowland Ltd has seen some major changes, we have seen 6 Kings ; George IV ; William IV ; Edward VII ; George V ; Edward VIII , George VI and 2 Queens, Victoria and currently Queen Elizabeth II rule our Country.

July 29th 2015
In a letter written to the seventh Earl of Shrewsbury in 1608, we read that upon visiting Sheffield, the writer thought he might be €œhalf choked with town smoke.€ The dregs of the industrial revolution are still seen today, all over the UK, in black soot coating on any building that still stands and was standing during that time period. But the history of the UK metal industry goes back much further than the industrial revolution.

July 22nd 2015
If you live in Yorkshire, you know that the metal industry is an integral part of this area. Besides the Victorian architecture and railways, this part of the UK is known for the role it plays in metal and the metal industry. Those in the know often call Yorkshire the metal capital of the world, but is it really?
Contact Us
Please fill in the short form below for any enquiries.
Search
Please fill in the short form below for any enquiries.